Government
California agency invests $4 million in stem cell treatment for Parkinson’s Disease
The California Institute for Regenerative Medicine (CIRM) is investing $4 million in a late-stage preclinical project by Ryne Bio aiming to improve treatment…


The California Institute for Regenerative Medicine (CIRM) is investing $4 million in a late-stage preclinical project by Ryne Bio aiming to improve treatment for Idiopathic Parkinson’s disease (PD).
PD is characterized by a loss of dopamine producing neurons that result in motor symptoms, such as dyskinesias (involuntary, erratic, writhing movements of the face, arms, legs or trunk) and non-motor effects such as dementia, depression and sleep disorders.
PD is the second-most common neurodegenerative disease after Alzheimer’s disease affecting approximately 1 million people in the U.S. In California, it is estimated that 116,900 people live with PD, representing the highest number of people with the disease in the country.
At its early stages, PD can be treated with medication such as Levodopa to treat symptoms but these become less effective as the disease progresses.
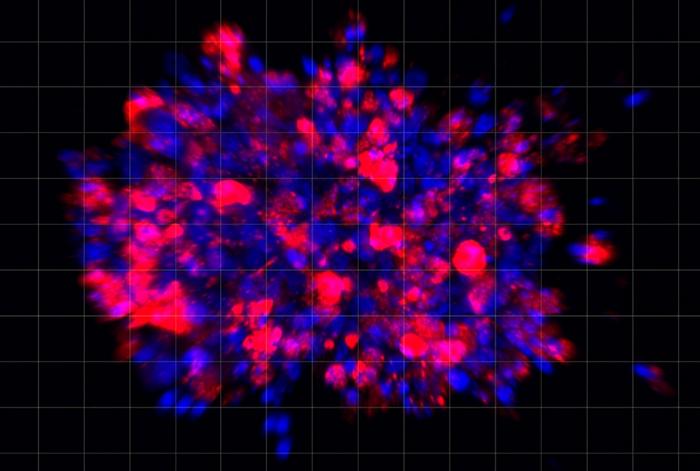
The proposed stem cell therapy in this project offers the potential to restore dopamine neurons, which play a role in many important body functions, including movement and memory.
Investigators at Ryne Bio are aiming to deliver dopamine producing cells to replace the lost neurons to the brain of Parkinson’s disease patients to restore/improve motor function.
The current grant is being funded to conduct Investigational New Drug (IND) enabling, nonclinical safety studies per the US Food and Drug Administration (FDA) Guidance. The IND is the authorization needed to begin a clinical trial in Parkinson’s patients.
CIRM has a vested interest in seeing this therapy succeed. To date, CIRM has invested more than $59 million in helping research for Parkinson’s disease progress from a basic or Discovery level through clinical trials.

Here Are the Champions! Our Top Performing Stories in 2023
It has been quite a year – not just for the psychedelic industry, but also for humanity as a whole. Volatile might not be the most elegant word for it,…
AI can already diagnose depression better than a doctor and tell you which treatment is best
Artificial intelligence (AI) shows great promise in revolutionizing the diagnosis and treatment of depression, offering more accurate diagnoses and predicting…
Scientists use organoid model to identify potential new pancreatic cancer treatment
A drug screening system that models cancers using lab-grown tissues called organoids has helped uncover a promising target for future pancreatic cancer…